The majority of cerebrovascular problems can be identified through diagnostic imaging tests. These allow neurosurgeons to view arteries and vessels in and around the brain and brain tissue itself. While technical progress has provided safe, fast, and more accurate diagnosis, it has also led to a wide variety of cerebrovascular imaging techniques from which one must choose. Optimal diagnosis and characterization of cerebrovascular disease requires selection of the appropriate imaging exam for each clinical situation.
Cerebrovascular Diagnostic Tests
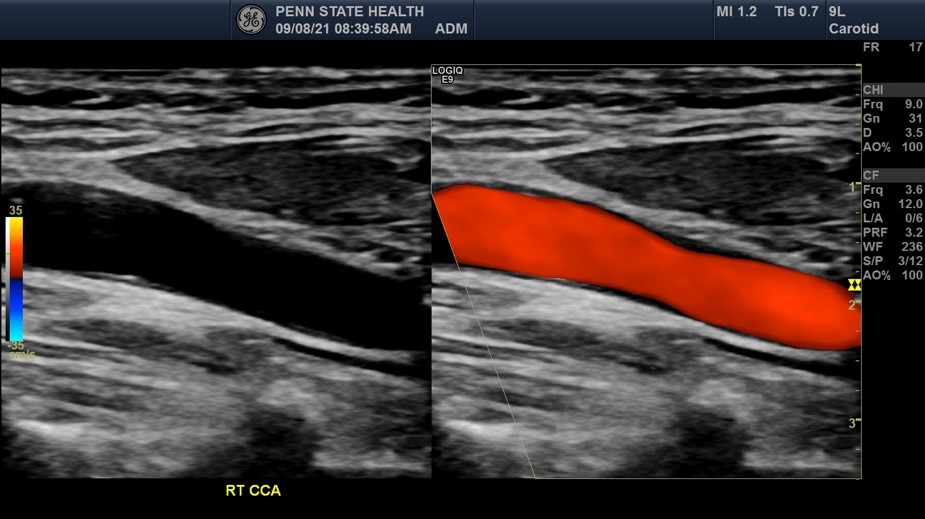
Carotid duplex (also called carotid ultrasound)
Ultrasound is a study that uses sound waves to ‘see’ inside your body. A carotid duplex ultrasound is performed to evaluate symptoms such as dizziness, loss of memory, stroke, loss of muscle control, and other symptoms that may result from the narrowing or blockage of the vessels on either side of your neck (carotid arteries). Additionally, carotid ultrasound can be used to screen for stroke risk.
A water-soluble gel is applied to your neck area with a transducer applied to each side of your neck. This gel helps transmit sound produced from a transducer to the skin surface. A transducer is a small, microphone-like device that uses sound waves to bounce off the organs and tissues in your body and the blood moving in your arteries, creating “echoes.” These echoes are reflected back to the transducer with a monitor showing images after converting the echoes to electronic signals that generate an image of the carotid arteries and pulse wave forms. There are no known risks from radiation, and this test is noninvasive and painless.
This test if often used to follow carotid stenosis. If the test shows an increase in the velocity of the blood moving through the narrowed artery, it could represent increased stenosis. Some surgeons find this definitive but others might want a more detailed type of scan before recommending surgery.
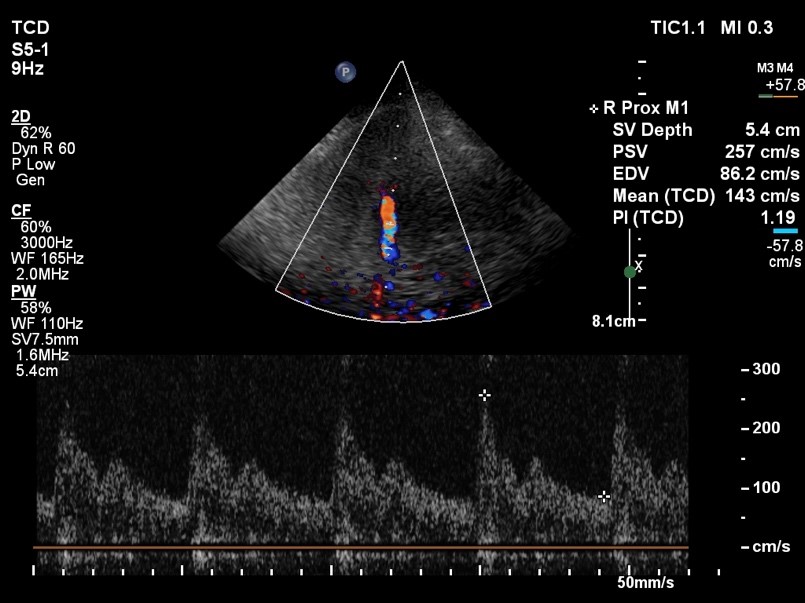
Transcranial doppler ultrasound (TCD)
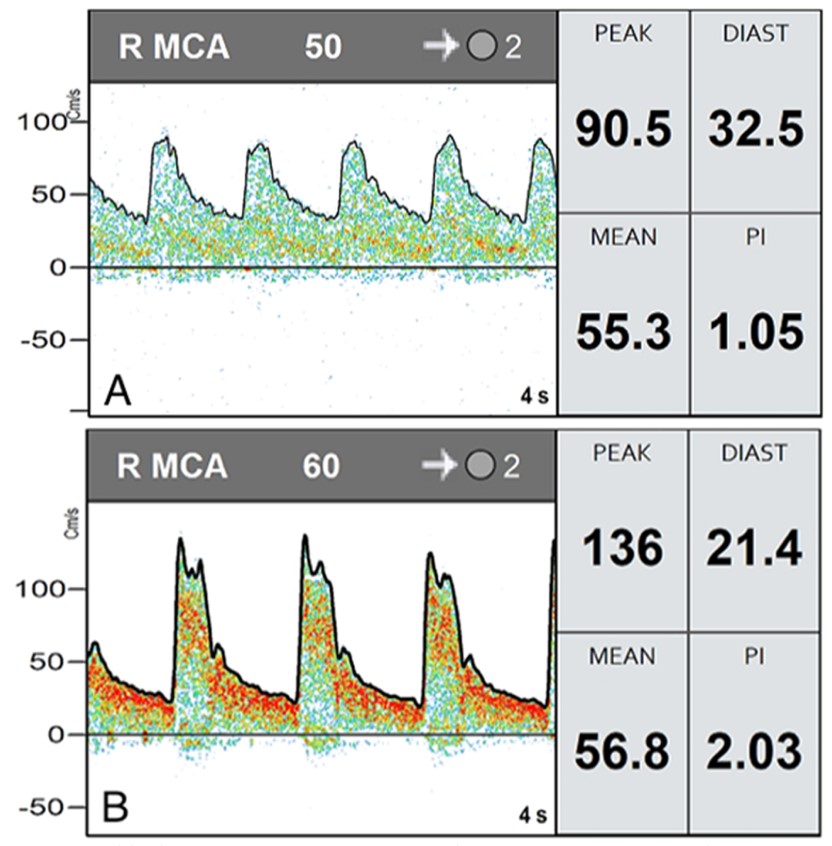
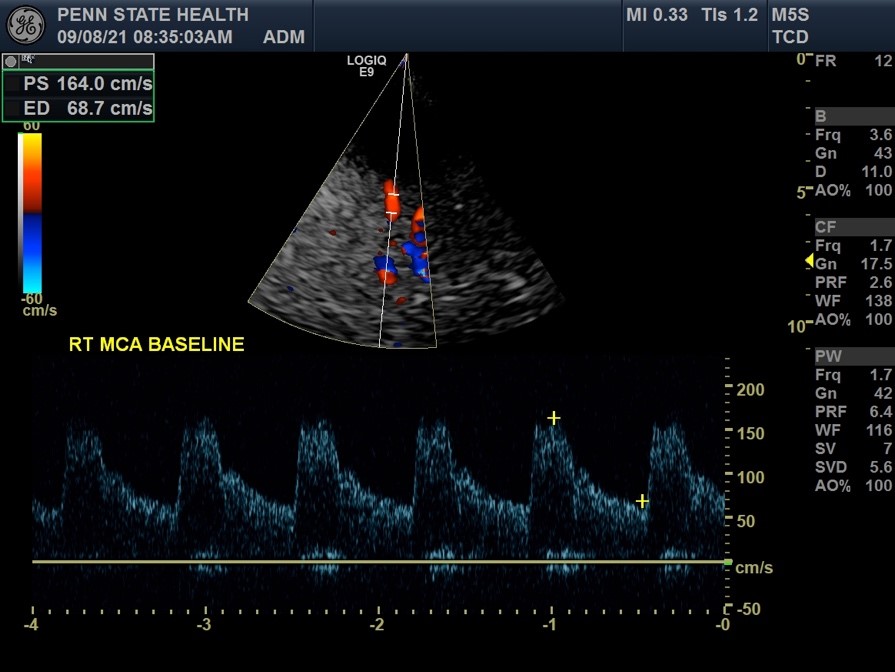
Transcranial doppler ultrasound uses sound waves to examine blood flow in your brain and is used to test for medical conditions that affects blood flow to and within the brain in addition to monitoring the results of certain treatments (e.g. the breakup of clots inside brain arteries).
A water-soluble gel is applied to the skin over the area to be examined (e.g. back of the neck, above the cheek bone, in front of the ear, over the eyelid). A transducer is held in place over the exam area, sending high-frequency sound waves through the brain. These sound waves are reflected off of blood cells moving within your blood vessels to capture information regarding blood flow in your brain (including speed and direction). The ultrasound signal is turned into graphs or color pictures that are shown on the display screen. There are no known risks from radiation, and this test is noninvasive and painless.
Sometimes a patient will have a very thick skull which prevents the probe from being able to acquire a strong enough signal to take a measurement or create a picture. Transcranial Doppler is often used daily in patients in the ICU after a ruptured aneurysm to evaluate for the development of cerebral vasospasm. If the values increase enough, further medical or endovascular treatment might be necessary.
In the outpatient setting, this test is sometimes used to determine if a narrowed artery leading to the brain is throwing emboli to the brain. The patient sits with the probe on his or her head for approximately an hour and any “blips” or “hits” are recorded. A neurologist or radiologist then reviews the recordings to determine if they demonstrate emboli or artifact.
Computed tomography (CT)
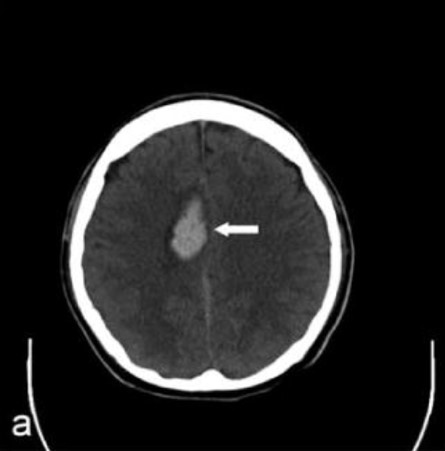
A noninvasive image that uses specialized X-ray measurements to produce more detailed information about brain tissue and structures than standard X-rays of the head, thus providing more data related to injuries and/or diseases of the brain. A CT of the brain may be performed to assess for tumors and other lesions, injuries, intracranial bleeding, structural anomalies (e.g. hydrocephalus, infections, brain function, or other conditions), particularly when other types of examinations are inconclusive. Other uses of brain CT is to provide guidance for brain surgery or biopsies of brain tissue as well as to evaluate the effects of treatment on brain tumors and to detect clots in the brain that may be responsible for strokes using medication that will be injected through a vein.
During a brain CT, an X-ray beam moves in a circle around the body, allowing many different views of the brain. The X-ray information is sent to a computer that interprets the X-ray data to display it in a two-dimensional (2D) form on a monitor. Scans may be done with or without “contrast.” Contrast refers to a substance taken by mouth or injected into an intravenous (IV) line that allows for a particular organ or tissue under study to be seen more clearly. Although a brain CT is noninvasive, there is radiation exposure and risks associated with this may be related to the cumulative number of X-ray examinations and/or treatments over time. Additionally, radiation exposure during pregnancy may lead to birth defects and special precautions should be used to minimize exposure to a fetus. Contrast media may cause allergic reactions, and patients who are allergic or sensitive to medications should notify their doctor. Sometimes special pre-medication can be given to contrast allergic patients to make this procedure safe in the setting of a pre-existing allergy.
For cerebrovascular surgery, plain ct (ie no contrast given) can provide a rapid and very accurate assessment of blood in the brain, which is usually scaled as a light gray and is clearly visible against the dark gray brain, bright white skull, or pitch black air. Plain CT can also show stroke or infarcted brain as much darker gray than healthy brain.
Computed tomography angiography (CTA)
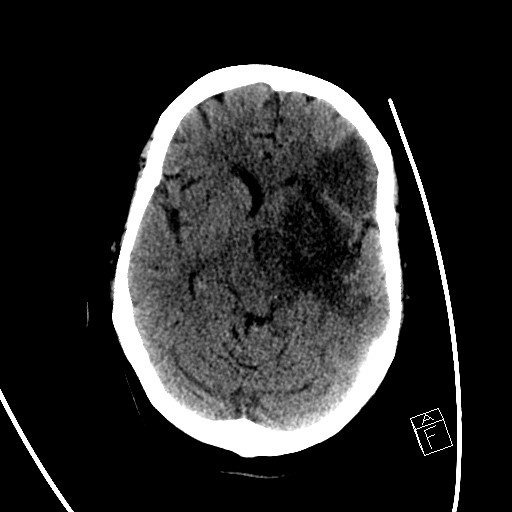
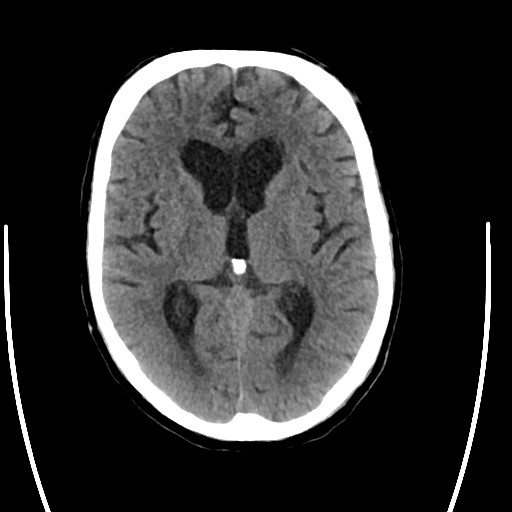
CT angiography (CTA) uses X-rays to visualize blood flow in arterial vessels throughout the body, from arteries serving the brain to those bringing blood to the lungs, kidneys, arms, and legs. Compared to catheter angiography (which involves the injection of contrast into an artery), CTA is much less invasive and contrast material is injected into a vein. This exam “lights up” blood vessels and tissues being studied and has been used to screen large number of individuals for arterial disease (e.g. aneurysm, atherosclerotic disease, abnormal blood vessel formations, damaged blood vessels due to injury, blood clots, and to evaluate tumors fed by blood vessels). Beams of X-rays are passed from a rotating device through the area of interest in the patient’s body from several different angles so as to create cross-sectional images, which are then assembled by a computer into a three-dimensional (3D) picture of the area being studied. Radiation exposure is a risk of CTA, as well as allergic reactions from contrast material and kidney damage if you have severe kidney disease or diabetes. If you are pregnant or breastfeeding, you should notify your healthcare provider and will need to wait for 24 hours following this test before nursing again.
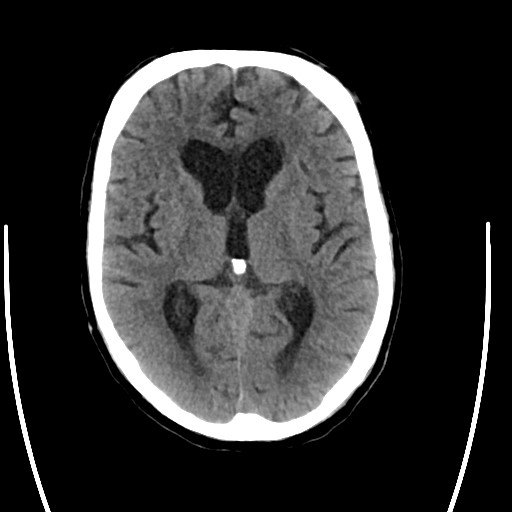
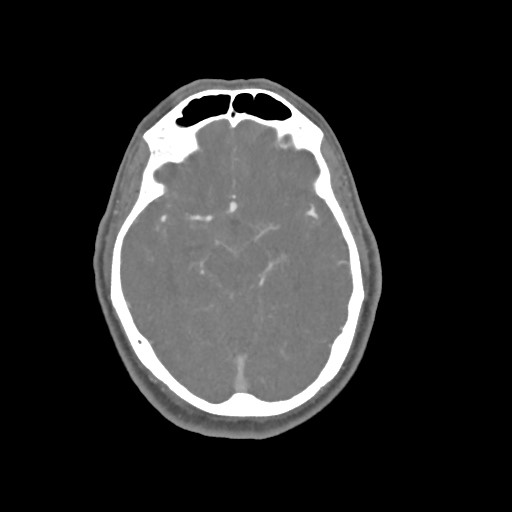
Computed tomography perfusion study (CTP)
CT perfusion studies in ischemic stroke has become an important adjunct, along with CT angiography (CTA), to conventional unenhanced CT brain imaging. CT perfusion studies enable differentiation of salvageable ischemic brain tissue (penumbra) from irreversibly damaged brain (infarct core). Therefore, this becomes useful when assessing a patient for potential treatment (e.g. thrombolysis or clot retrieval). Other potential uses of CT perfusion imaging that has been identified has been in evaluating and following chronic cranial and extracranial atherosclerotic-occlusive disease, assessing vessel spasm after subarachnoid hemorrhage, distinguishing brain cancer from infections, and in confirming brain death.
CT perfusion is based upon a tracer kinetic model (e.g. CT perfusion, MR perfusion) and assumes a nondiffusible tracer. This first-pass technique monitors changes in density as a function of time. Pixel-based time attenuation curves are then produced by deconvolution. From this data, quantitative cerebral perfusion maps, including cerebral blood flow, cerebral blood volume, and mean transit time, are constructed. In other words, the CT scan takes X-ray pictures over time as the body takes up more and more of a nonradioactive substance that the cells of the body take in. CT perfusion can be performed quickly, safely, and in the same scanner used for a noncontrast brain CT. Therefore, CT perfusion studies do expose the patient to a small amount of radiation.
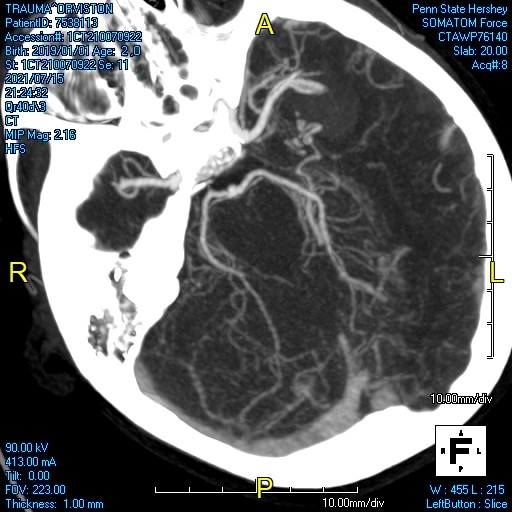
Magnetic Resonance Imaging (MRI)
A noninvasive technique that uses magnetic field and computer-generated radio waves to create detailed images and is the most frequently used imaging test of the brain and spinal cord. Unlike X-rays or computed tomography (CT scans), MRI does not use ionizing radiation. MRI may be preferable to CT scans in situations where organs or soft tissue is being studied, as it is better at telling the difference between normal and abnormal tissue. MRIs may be used to examine the brain for injuries or the presence of structural abnormalities or other conditions including tumors, abscesses, congenital abnormalities, aneurysms, venous malformations, hemorrhage, and hydrocephalus.
An MRI machine is a large, cylindrical (tube-shaped) machine that creates a strong magnetic field around the patient. This field, along with radio waves, alters hydrogen atoms natural alignment in the body and the pulses from these radio waves (produced by the scanner) knock the nuclei of your atoms out of normal position. As the nuclei realign back into their proper position, they send out radio signals which are received by a computer to analyze and convert them into a two-dimensional (2D) image of the brain. Although the test is non-invasive, some patients may experience claustrophobia in the imager.
The powerful magnetic field can affect implanted metal and therefore patients with gunshots or surgical implants like artificial joints, pacemakers, or stimulators may not be able to have an MRI. Some programmable VP Shunts are MRI safe but need to be reprogrammed after each scan. All modern endovascular devices are MRI compatible but certain older surgical clips might not be. Often technologists will ask for a make and model of any devices so it is helpful to have this information before you go for a scan.
MRI is very sensitive for the swelling that goes along with a cerebral infarction or ischemic stroke and can identify strokes earlier than CT and strokes that are too small for a CT.



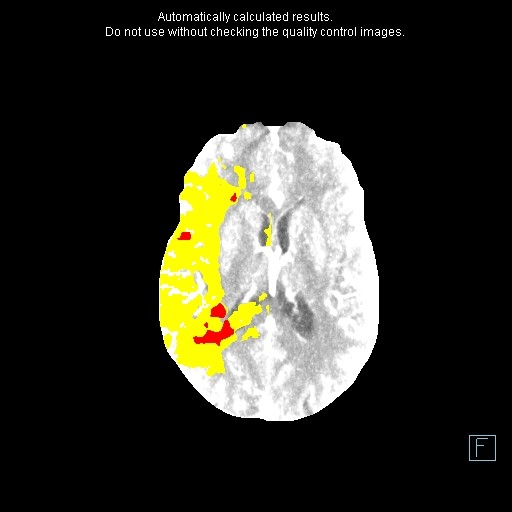
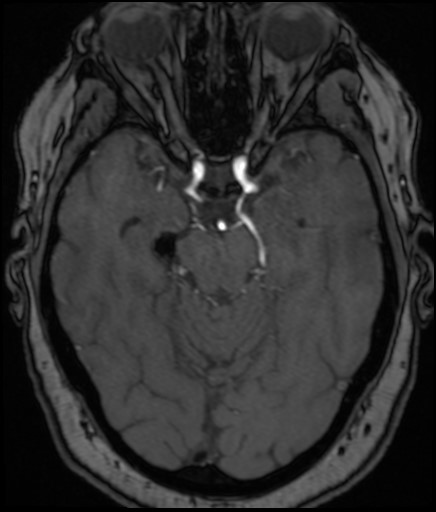
Magnetic Resonance Angiogram (MRA)
Magnetic resonance angiography (MRA) is a type of MRI that looks specifically at the brain’s blood vessels. Unlike catheter angiography, which requires inserting a catheter into an artery, MRA is noninvasive and less painful. Unlike a CTA, no ionizing radiation is used. Furthermore, an MRA does not necessarily require contrast and if it does it is not iodine based but is gadolinium.
MRA is particularly helpful at evaluating coiled aneurysms.
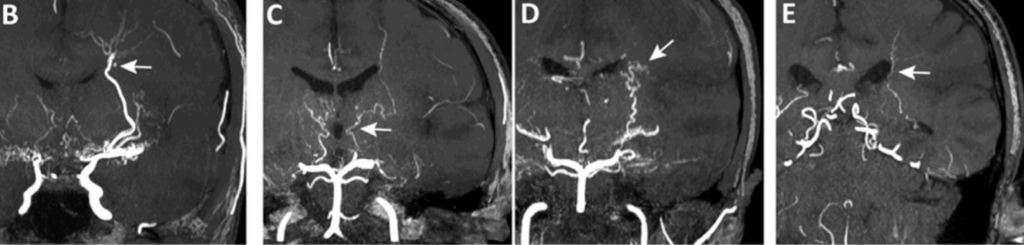
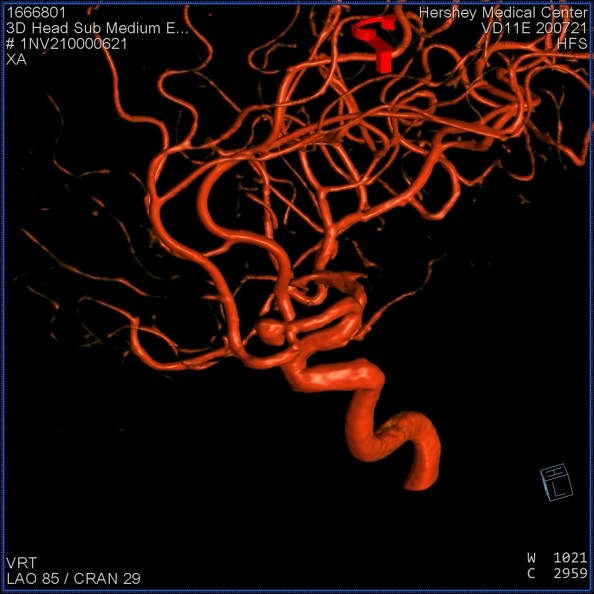
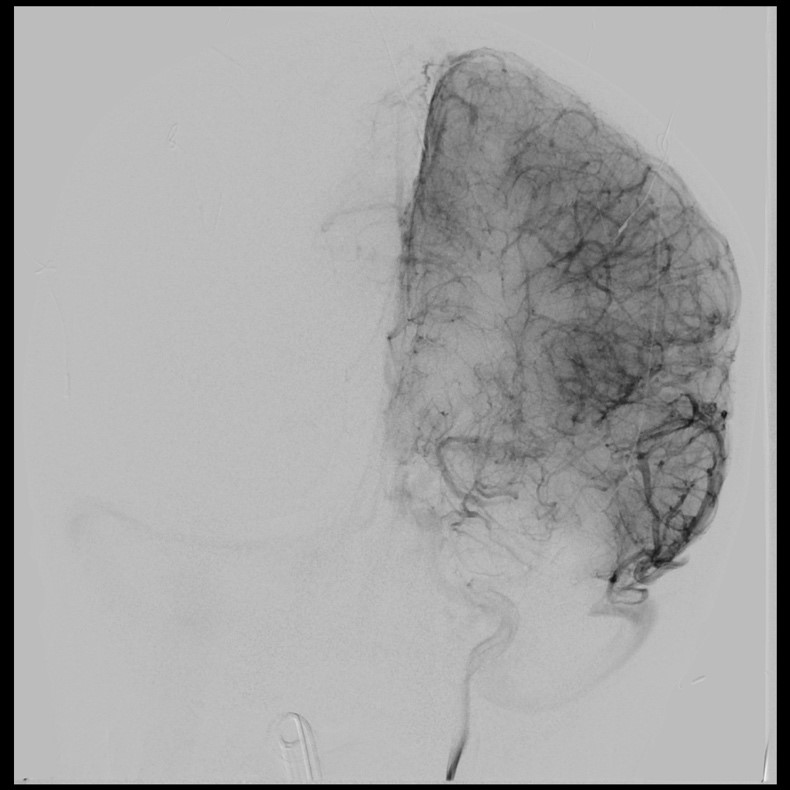
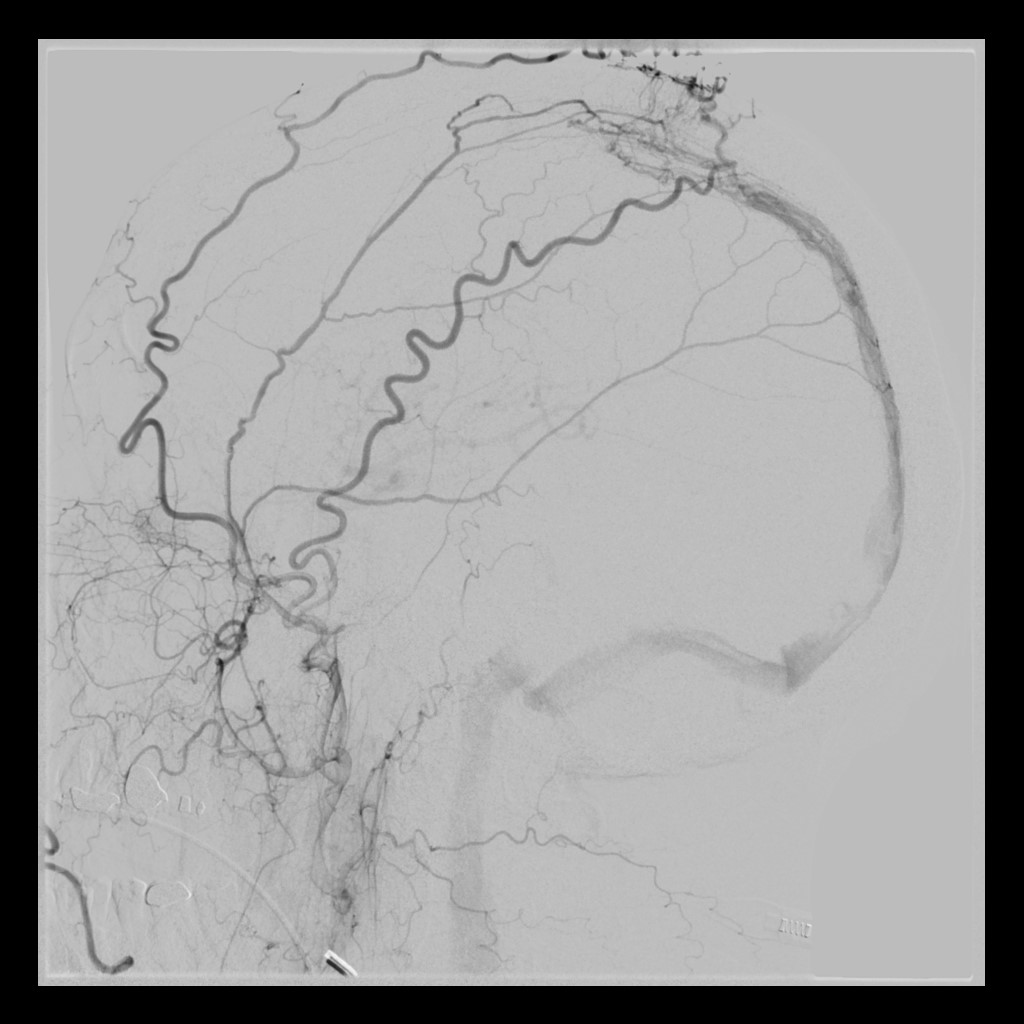
Cerebral Angiography (also called vertebral angiogram, carotid angiogram)
Cerebral angiography, also known as intra-arterial digital subtraction angiography, is an invasive procedure that produces X-ray pictures of the blood vessels in the brain and neck. Specifically, this test looks for changes in the blood vessels including blockages (occlusive disease), narrowing (stenosis), ballooning (aneurysm), inflammation (vasculitis), vessel spasm (vasospasm), and vascular malformations.
In this procedure, contrast dye is utilized as arteries and veins are not normally seen in an X-ray picture. The patient is given a local anesthetic, the artery is punctured (usually in the leg) and a needle is inserted into the artery. A catheter (a long, narrow, flexible tube) is inserted through the needle and into the artery. It is threaded through the main vessels of the abdomen and chest (aorta) until it is properly placed in the arteries of the neck and head. This procedure is monitored by a fluoroscope (a specialized X-ray that is projected onto a TV monitor to verify the location of the catheter and to monitor the vessels of the head and neck after injecting contrast material). Contrast dye is injected via the catheter to make blood vessels show up on the X-ray image and allows the healthcare provider to better see the structure of the vessels, at which time a series of X-rays will be taken (of the arteries, capillaries, and veins). During the procedure and contrast dye injection, you may feel a flushing sensation, a salty or metallic taste in your mouth, a brief headache, and nausea and/or vomiting. When the test is done, the catheter is removed and pressure is applied over the artery to prevent bleeding and formation of a hematoma (a collection of blood under the skin). The risks to this procedure includes radiation exposure and risk of allergic reaction to contrast dye. As this procedure is invasive and involves the blood vessels and blood flow of the brain, there is a small risk of transient ischemic attack (brief-stroke like condition), stroke, bleeding, blood clots, paralysis on one side of the body (hemiplegia), loss of the ability to speak or understand speech (aphasia), and hematoma at the catheter placement site. In experienced hands, the risk if very low.
This study provides the gold standard look at cerebral blood vessels with a level of detail beyond that of the other modalities. Furthermore, since pictures are taken multiple times per second, blood flow can be evaluated. This is particularly helpful in AVMs and AVFs.
The procedure usually takes about 45 minutes and often a patient has to lay flat or stay in the hospital recovery area for a few hours after. Most facilities ask that an angiogram patient not drive themselves home.
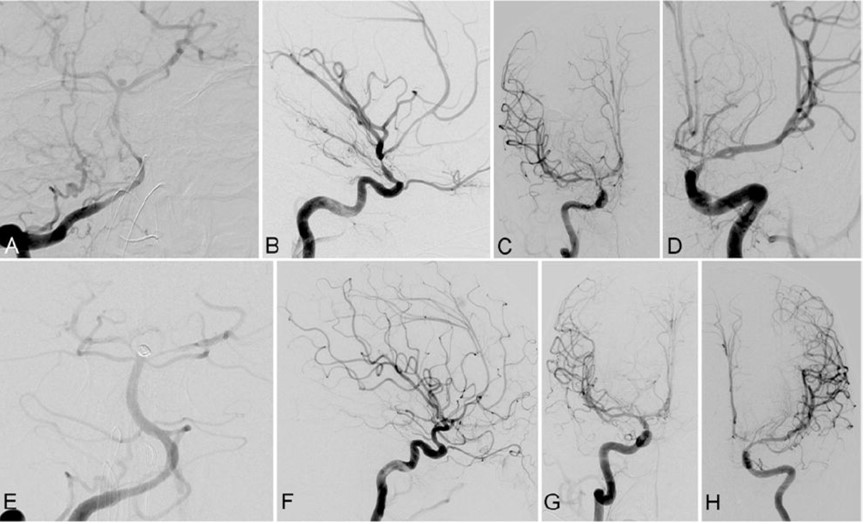
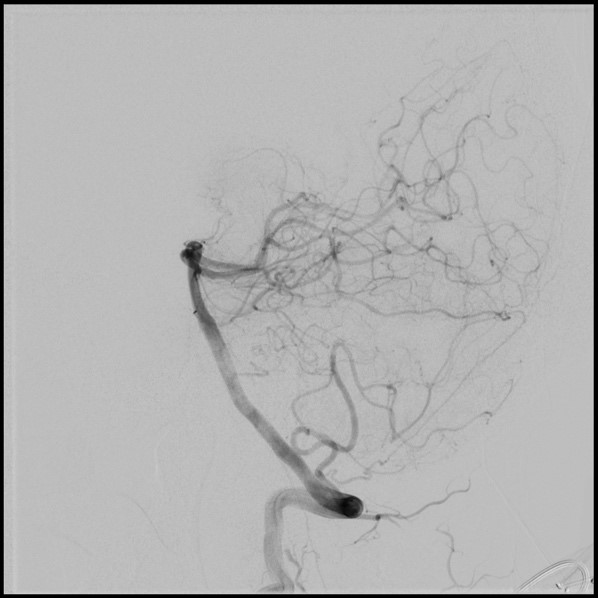
Author Information
Michael Gigliotti, MD; Scott Simon, MD, FAANS
Penn State College of Medicine at the Milton S. Hershey Medical Center, Hershey, Penn.
Note from AANS
The AANS does not endorse any treatments, procedures, products or physicians referenced in these patient fact sheets. This information is provided as an educational service and is not intended to serve as medical advice. Anyone seeking specific neurosurgical advice or assistance should consult his or her neurosurgeon, or locate one in your area through the AANS’ Find a Board-certified Neurosurgeon online tool.
