Overview
Craniopharyngiomas are histological benign, but locally aggressive, tumors that develop near the pituitary gland at the base of the brain. Although malignant transformation of craniopharyngioma has been reported, this is a rare event. These tumors usually involve the pituitary stalk, the structure that connects the pituitary gland to the hypothalamus and are classically very sticky and adherent tumors. They have varying cystic and solid portions, with the majority having at least some cystic component.
They are very rare, with a prevalence of roughly two per 1,000,000 people, and account for approximately 2-3% of all brain tumors. They exhibit no gender or racial bias and commonly present during childhood (ages 5-14 years) or late adulthood (ages 50-74 years).
SYMPTOMS + TYPES
Presenting symptoms depend on the location and size of the tumor. Some tumors can be large before they cause symptoms, while others can present while very small. Symptoms include:
- Headache
- Peripheral field vision loss
- Eye movement weakness/double vision
- Vomiting
- Personality changes, confusion
- Hormonal insufficiency/hypothalamic dysfunction
- Headache
- Peripheral field vision loss
- Eye movement weakness/double vision
- Vomiting
- Personality changes, confusion
- Hormonal insufficiency/hypothalamic dysfunction
- Adrenal insufficiency
- Fatigue
- Low blood pressure
Risk Factors
Craniopharyngiomas are sporadic tumors, meaning the cause of their development is unknown and there are no known risk factors.
Diagnostic Imaging
Following a thorough history and physical examination by a doctor, additional tests may be ordered for a patient showing signs of craniopharyngioma. These include imaging studies, such as magnetic resonance imaging (MRI) and computed tomography (CT).
MRI: An MRI is typically performed with the administration of intravenous dye to identify structures within the brain and evaluate for a tumor such as a craniopharyngioma. This helps providers identify the location of important brain structures that are located near the tumor, including the hypothalamus, pituitary stalk and gland, internal carotid arteries, optic nerve and optic chiasm. Several classification systems of craniopharyngiomas are based on the imaging findings and may help elucidate the best treatment option.
CT: A CT scan can be performed to evaluate for calcifications within the craniopharyngioma.
Types of Craniopharyngiomas
- Adamantinomatous Variant: Histopathological findings include wet keratin, calcifications, stellate reticulum and palisading epithelium. Mutations within exon 3 of CTNNB1, a gene encoding beta-catenin, have been noted.
- Papillary Variant: This subtype typically lacks calcifications and is almost exclusively found in adults. Mutations within exon 15 of BRAF V600E have been identified. Ongoing research investigating the effectiveness of medical therapy with BRAF inhibitors for this subtype has begun with encouraging preliminary results.
TREATMENT + CARE
Surgery
Surgical resection of a craniopharyngioma is the preferred initial treatment for most patients. This establishes and confirms the specific tumor and histological subtype diagnosis, provide an opportunity to remove the pressure the craniopharyngioma may exert on normal brain structures such as the optic nerves and optic chiasm and possibly improve vision, as well as offer the best chance to remove as much of the tumor as possible. Portions of tumors adherent to the hypothalamus are usually not resected, because of the potential devastating effects of hypothalamic injury. There are some risks with surgical intervention, so it is important to discuss them thoroughly with an experienced neurosurgeon.
There are two basic categories of surgical approaches that each have a distinct set of advantages and disadvantages. The neurosurgeon’s experience, the patient’s age and overall health, as well as the size and location of the craniopharyngioma influence the particular approach and surgical plan.
- Open craniotomy: The neurosurgeon makes an incision on the scalp or eyebrow region, temporarily removes a window of bone, opens the lining of the brain and removes the tumor usually with the aid of an operating microscope. The bone is usually then replaced with small plates and screws and the incision is sewn back together.
- Endonasal (or transphenoidal): The neurosurgeon often works with an otolaryngologist who provides access to the skull base through the natural nasal passageway. This approach is typically performed with an endoscope, which is a small camera and light source on a shafted instrument. The neurosurgeon then removes the tumor and recreates the separation between the brain and nasal pathway with varying types of tissue, including nasal mucosa, abdominal fat, thigh tissue, collagen grafts and surgical glue.
Case example: Endoscopic Endonasal Resection of 6cm Craniopharyngioma
A 58-year-old female presenting with 40 years of blindness and loss of some hormonal function. She has a history of surgery for a craniopharyngioma at an outside hospital roughly 40 years prior to her presentation. Imaging revealed a nearly 6 cm recurrent craniopharyngioma with solid and cystic components. She underwent an endoscopic endonasal approach with complete removal of the tumor and skull base reconstruction with a pedicled vascularized nasoseptal flap. Following surgery, her vision remained stable, and she did not require any new hormonal therapy upon leaving the hospital.
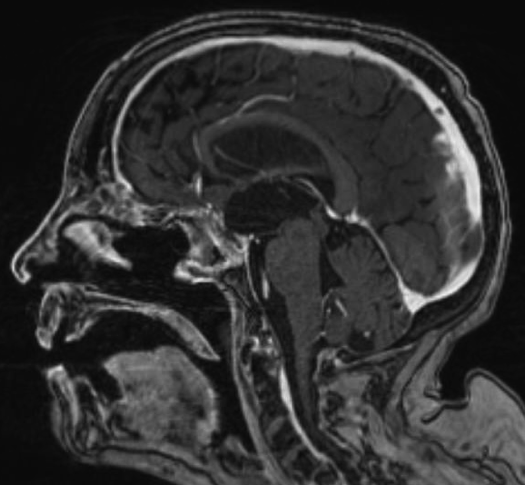
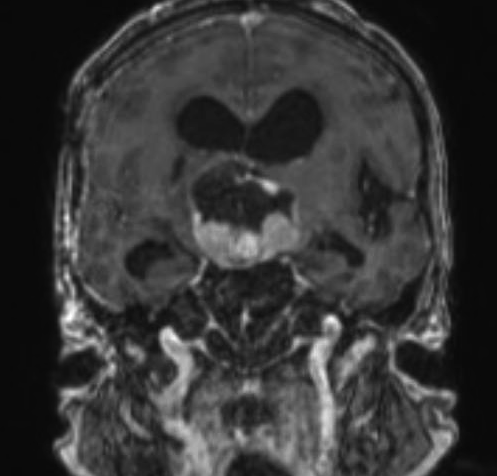
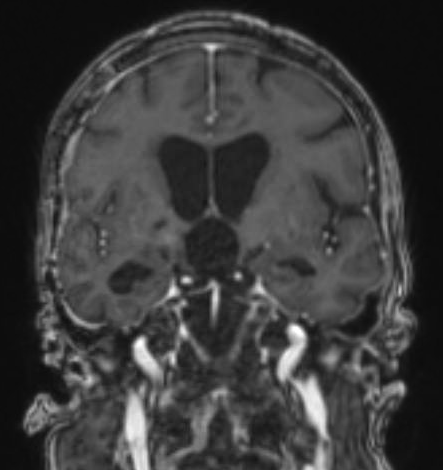
Other Treatments
- Radiotherapy: Radiation works by damaging the DNA of the tumor and can be administered through different techniques (standard external beam, stereotactic radiosurgery and proton beam). With craniopharyngiomas, it can be used following surgical resection to target any residual or recurrent tumor and in treating tumors adherent to the hypothalamus. There are risks with radiotherapy that need to be discussed in detail with the provider.
- Miscellaneous: There is literature that discusses less common, as well as emerging, treatments for craniopharyngiomas. This includes the use of chemotherapy, such as paclitaxel, carboplatin and the administration of BRAF inhibitors for papillary craniopharyngioma, surgical placement of intracavitary phosphorus-32, yttrium, bleomycin, rhemium and the application of interstitial brachytherapy. There are risks with these treatment options that need to be discussed in detail with the provider.
SOURCES
- Müller, H. L. (2014). Craniopharyngioma. Endocrine Reviews, 35(3), 513–543. https://doi.org/10.1210/er.2013-1115
- Garnett, M. R., Puget, S., Grill, J., & Sainte-Rose, C. (2007). Craniopharyngioma. Orphanet Journal of Rare Diseases, 2(1), 18. https://doi.org/10.1186/1750-1172-2-18
- Garrè, M. L., & Cama, A. (2007). Craniopharyngioma: modern concepts in pathogenesis and treatment. Current Opinion in Pediatrics, 19(4), 471–479. https://doi.org/10.1097/mop.0b013e3282495a22
- Theodosopoulos, P. V., et al. (2012). Schmidek and Sweet: Operative Neurosurgical Techniques (6th ed.). Saunders.
- Lo W.B., et al. (2019). Craniopharyngioma. Complications in Neurosurgery.
- Kan, P., Duckworth, E., Germanwala, A. V., Pelargos, P., Cho, J. M., Choy, W., Smith, Z. A., & Yang, I. (2015). Neurosurgery concepts: Key perspectives on embolectomy for stroke with emergent large vessel occlusion (MR CLEAN), endonasal endoscopic craniopharyngioma resection, gamma knife radiosurgery for meningiomas, therapeutic hypothermia for severe traumatic brain injury. Surgical Neurology International, 6(1), 165. https://doi.org/10.4103/2152-7806.168064
- Jusue-Torres, I., Hulbert, A., Germanwala, A. A., Patel, C. R., & Germanwala, A. V. (2018). The 100 Most-Cited Reports About Craniopharyngioma. World Neurosurgery, 119, e910–e921. https://doi.org/10.1016/j.wneu.2018.08.004
- Brastianos, P. K., & Santagata, S. (2016). ENDOCRINE TUMORS: BRAF V600E mutations in papillary craniopharyngioma. European Journal of Endocrinology, 174(4), R139–R144. https://doi.org/10.1530/eje-15-0957
- Kassam, A. B., Gardner, P. A., Snyderman, C. H., Carrau, R. L., Mintz, A. H., & Prevedello, D. M. (2008). Expanded endonasal approach, a fully endoscopic transnasal approach for the resection of midline suprasellar craniopharyngiomas: a new classification based on the infundibulum. Journal of Neurosurgery, 108(4), 715–728. https://doi.org/10.3171/jns/2008/108/4/0715
Note from AANS
The AANS does not endorse any treatments, procedures, products or physicians referenced in these patient fact sheets. This information is provided as an educational service and is not intended to serve as medical advice. Anyone seeking specific neurosurgical advice or assistance should consult his or her neurosurgeon, or locate one in your area through the AANS’ Find a Board-certified Neurosurgeon online tool.
